Sinopharm Vaccine European Medicines Agency - Ema Updates Covid 19 Vaccine Risk Management Plans To Reflect Variant Safety Raps
Thus far EMA has authorized the use of Comirnanty Moderna Vaxzevria and Jannsen vaccines. EMA is analysing data from vaccination campaigns and ongoing studies as soon as they become available to ensure the vaccines are used as safely and effectively as possible.

Ema Receives Application For Conditional Marketing Authorisation Of Covid 19 Mrna Vaccine Bnt162b2 European Medicines Agency
No decision has been made towards Sinopharm although it has already been purchased by some EU member countries.

Sinopharm vaccine european medicines agency. However the European Medicines Agency EMA has not yet reviewed it for use in the European Union. The European Medicine Agency has started a rolling review of the Covid-19 vaccine developed by Chinese company Sinovac the latest vaccine candidate hoping to. The European Commission has authorised several vaccines to prevent COVID-19 disease in the European Union EU following evaluation by the European Medicines Agency EMA.
Increase in vaccine manufacturing capacity for COVID-19 vaccines from BioNTech Pfizer and Moderna. According to the European Medicines Agency once the vaccine is administered the modified virus enters the bodys cell and delivers the genes of the SARS-CoV-2 spike protein. The Sinopharm Vaccine or BBIBP-CorV has been developed by the Beijing Bio-Institute of Biological ProductsBBIBP in collaboration with the Sinopharm Vaccine or BBIBP-CorV.
The prime minister has rejected arguments that only vaccines approved by the European Medicines Agency the EUs medicines regulator should be approved for use in Hungary. In the UAE US-regulated Pfizer and Chinese-regulated Sinopharm are the most-widely used vaccines which raises questions about when and if Sinopharm will be approved for use in the UK travel programme. EMA is not involved in advising on travel requirements in the EU such as vaccination quarantine or testing for travellers.
Last week Hungarian authorities became the first in the EU to approve the Russian Sputnik V vaccine and jumped ahead of the European Medicines Agency in approving the AstraZeneca jab. He said that acceptable vaccination certificates are those where the holder has completed the vaccination scheme with one of the approved vaccines of the European Medicines Agency or the Russian Sputnik or the Chinese Sinopharm and the specified by the Ministry time since the last administration has passed depending on the date of travel. Another batch of Sinopharm COVID-19 vaccine arrived on August 2021 consisting.
Neither Chinas Sinopharm or Russias Sputnik V. EMA is not involved in advising on travel requirements in the European Union EU such as vaccination quarantine or testing for travellers. Still Hungarian officials expect that the Sinopharm vaccine which received final approval last week will provide a sharp boost in the countrys vaccination rate.
Vaccines authorised in the European Union EU to prevent COVID-19 following evaluation by the European Medicines Agency EMA. However the Sinopharm is yet to be approved by the European Unions health regulator agency European Medicines Agency. Elsewhere in the EU Hungary in February became the first country in the bloc to start using Sputnik V and Chinas Sinopharm vaccine neither of which has been approved by the European Medicines.
The FDA announced that three vaccine manufacturers namely PfizerBioNTech AstraZeneca and Sinovac have inquired on the process of obtaining an EUA in the Philippines. Sinopharm and the BBIBP opted to use. The imminent World Health Organisation WHO approval of Chinas Sinopharm vaccine will be a welcome relief for Middle East travellers and European economies alike according to Bruno Trenchard senior manager Hotels and Hospitality CBRE Middle East as the EU prepares to potentially ease restrictions on non-essential travel from overseas.
Vaccination with one of the approved vaccines of the European Medicines Agency EMA or with the Sputnik V vaccine or with the Chinese Sinopharm vaccine BBIBP COVID-19 and the specified by the Ministry of Health number of days has elapsed between the administration of the last dose depending on the vaccine administered. EMAs human medicines committee CHMP has adopted recommendations that will increase manufacturing capacity and supply of COVID-19 vaccines in the EU. European Medicines Agency Domenico Scarlattilaan 6 1083 HS Amsterdam The Netherlands.
According to a new tool developed by VisaGuideWorld which helps travellers to find out whether the country they plan on visiting accepts their COVID-19 vaccine as valid proof it has been revealed that the three vaccines that are most recognised in EU countries after the EMA ones are Covishield Sinopharm and Sinovac. The adenovirus is engineered to disable replication but the cell can still read the instruction in order to produce the spike protein. The vaccine was given authorization by Chinas National Medical Products Administration on 31st December 2020 while it has been given authorization for its emergency use.
European Medicines Agency European Union Vaccine manufacturers applied for EUA. The EU has also announced they will allow American tourists this summer as the US is using vaccines approved by the European Medicines Agency EMA. A UK Department of Health spokesperson confirmed to Arabian Business that Britain is only recognising vaccines approved by the UKs Medicines.
Decisions about which COVID-19 vaccines are included for example in the EU Digital COVID Certificate are taken by the EU Member StatesEMA is in charge of the scientific evaluation of vaccines for EU marketing authorisation.

Travellers Vaccinated With Russian Chinese Indian Vaccines May Be Unable To Enter Majority Of Eu Countries Schengenvisainfo Com

In Europe Union Vaccines From Russia And China Are Now Under Study The Washington Post
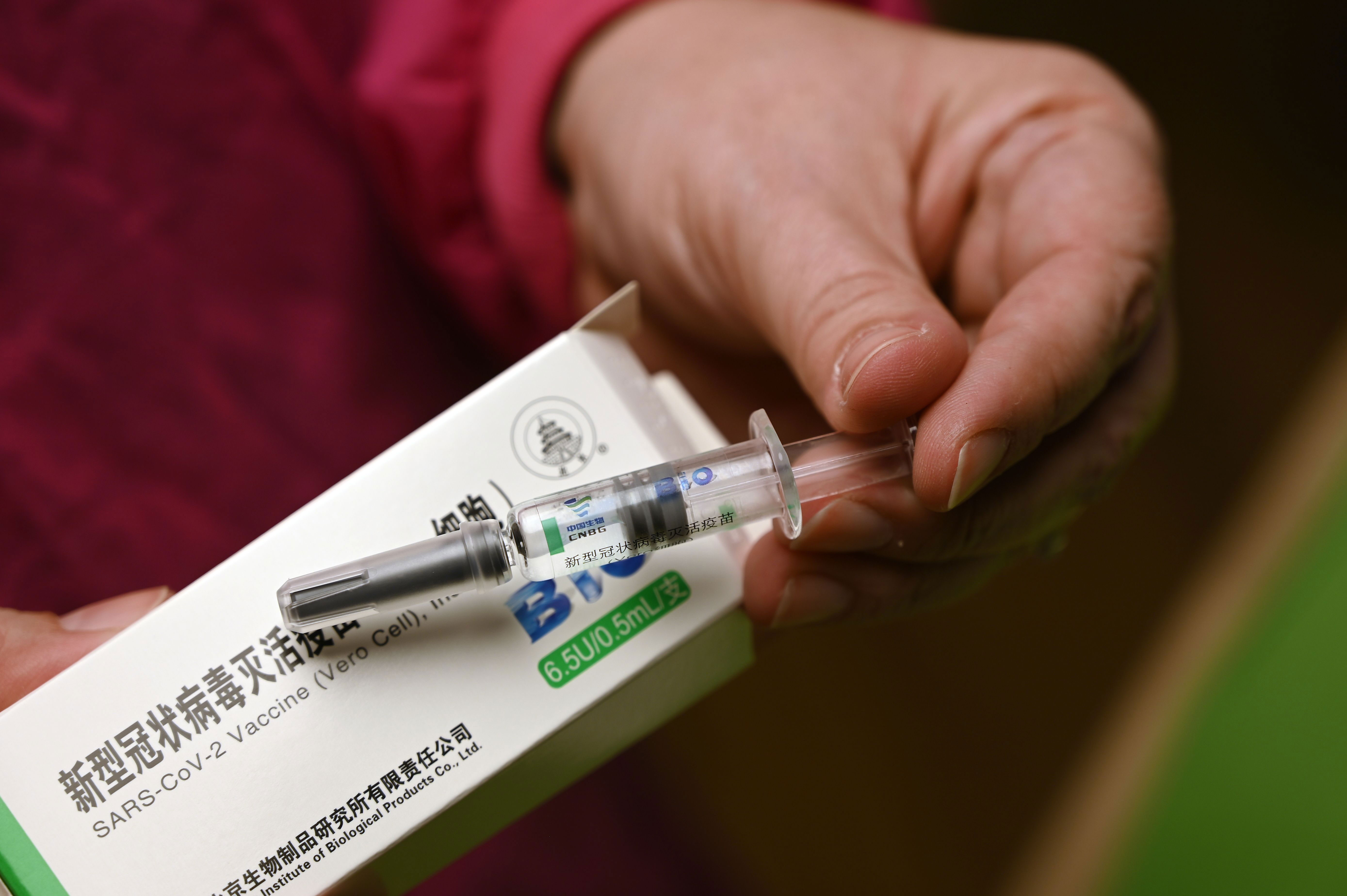
The Czech Republic Becomes The Second Eu Country To Ask For China S Sinopharm Vaccine

Eu Medicines Agency On Twitter Check Out Our Latest Updates On The Safety Of All Covid19vaccines Authorised In The Eu Comirnaty Https T Co Fszhe7c1rm Covid 19 Vaccine Janssen Https T Co 43n57rqa9t Spikevax Https T Co Rchzynkxsc

The Latest European Medicines Agency Deciding On Third Dose National News Santamariatimes Com
Eu Slams Russia And China For Western Vaccines Disinformation Campaign Euronews

Eu Medicines Agency On Twitter Ema Has Approved Increasing The Manufacturing Capacity Of Vaccines From Biontech Pfizer Moderna This Is Expected To Support The Continued Supply Of Covid19vaccines In The Eu

Eu Regulators Start Review Of China S Sinovac Vaccine

Ema Updates Covid 19 Vaccine Risk Management Plans To Reflect Variant Safety Raps
European Commission Gives Final Approval To Astrazeneca Covid 19 Vaccine Amid Distribution Dispute

Who And Ema Approved Vaccine List Which Covid 19 Vaccines Are On Who And Ema List Updated 30 August 2021 Wego Travel Blog

Travellers Vaccinated With Russian Chinese Indian Vaccines May Be Unable To Enter Majority Of Eu Countries Schengenvisainfo Com

Vaccines Approved And Under Development Against Covid 19

Eu Recognises Covid Certificates From Turkey And Ukraine Euractiv Com

Eu Medicines Agency On Twitter Ema S Third Regular Press Conference On Covid19 Vaccines And Treatments Will Be Broadcast Live Today June 17 From 16 00 Cest Https T Co Yu1ob7fhcr Emapresser Https T Co 2mcpjzl7lj
Doubts Mount About Efficacy Of Russia S Sputnik Vaccine Voice Of America English
In Eu First Sinopharm S Coronavirus Vaccine Approved By Hungary Nikkei Asia

Russia And China Have Not Sought The European Medicines Agency S Authorisation For Covid 19 Vaccines Europe News Top Stories The Straits Times
The Latest European Medicines Agency Deciding On Third Dose The Durango Herald

