Sinopharm Vaccine European Approval / More Eu Countries Eyeing Separate Deals With Russia For Sputnik Vaccine Politico
The sinopharm vaccine which has been readily used in the UAE Bahrain and other Middle East countries is awaiting official approval from the WHO. EMA is not involved in advising on travel requirements in the European Union EU such as vaccination quarantine or testing for travellers.

Why Who Approval Of Sinopharm Vaccine Will Boost European Coffers From Middle East Travel Arabianbusiness
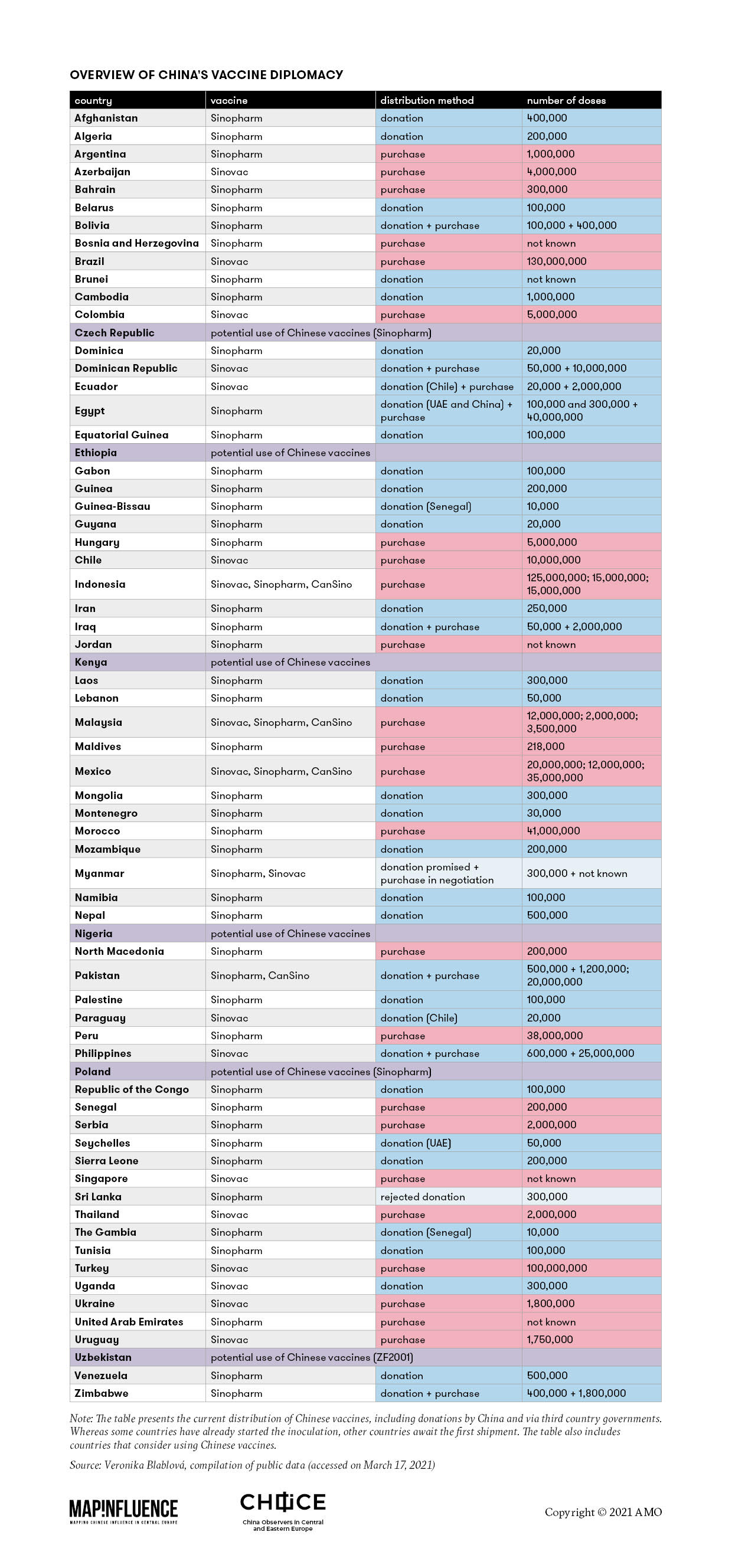
A number of African Latin American and Asian countries have chosen to approve and use the Vero Cell vaccine by the Beijing-based pharmaceutical company.
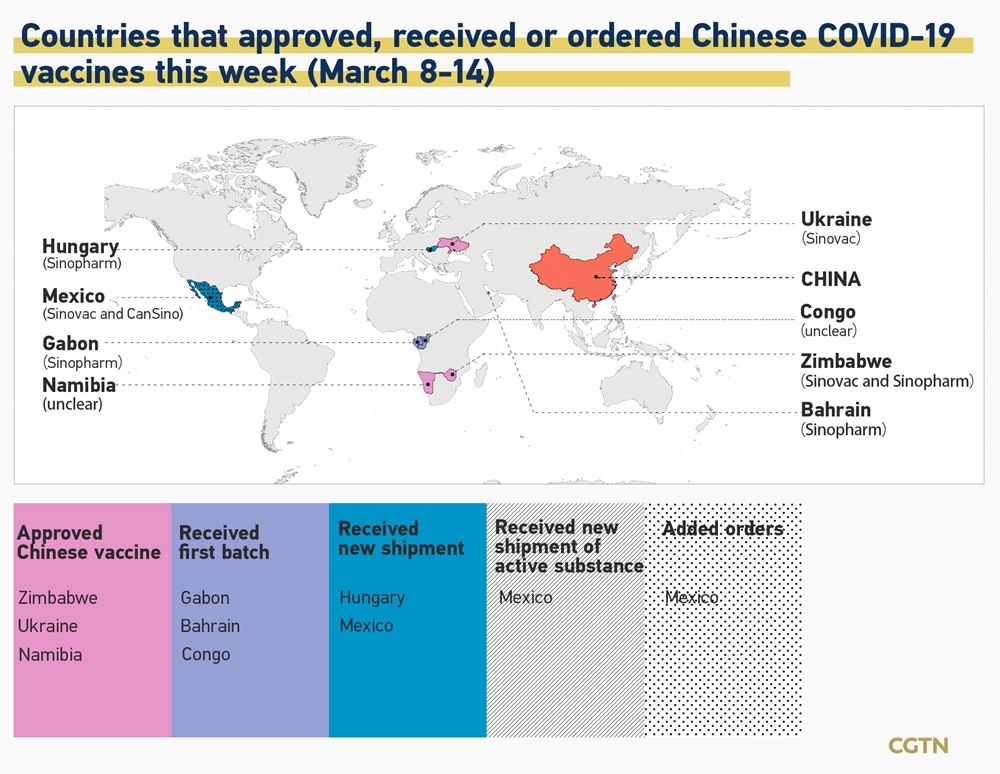
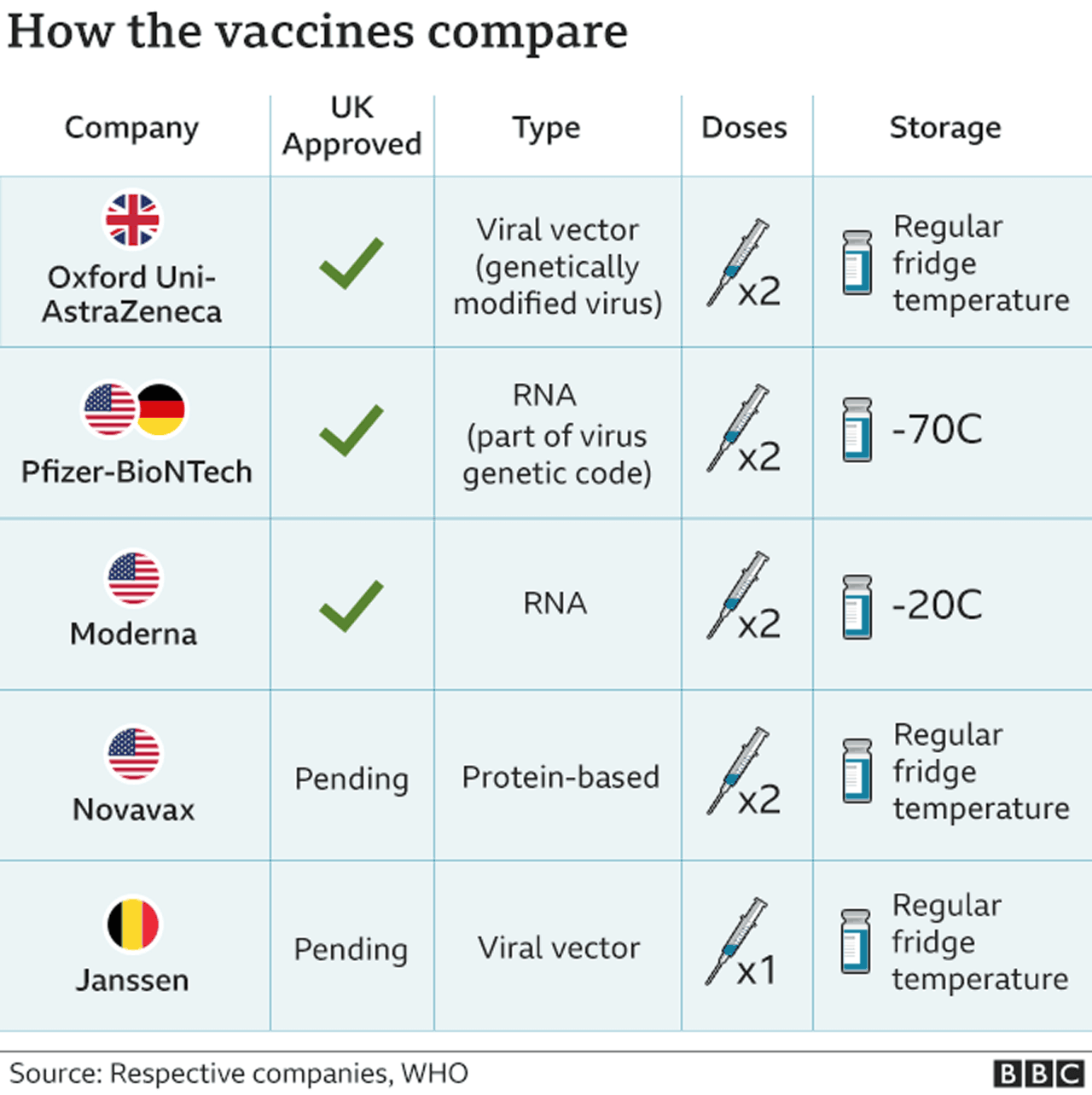
Sinopharm vaccine european approval. National regulatory authorities have granted emergency use authorizations for twenty-two COVID-19 vaccines. Six of those have been approved for emergency or full use by at least one WHO -recognized stringent regulatory authority OxfordAstraZeneca Pfizer-BioNTech Sinopharm-BBIBP Moderna Sinovac and Janssen. Why WHO approval of Sinopharm vaccine will boost European coffers from Middle East travel.
WHO approves Sinopharm vaccine boosting Gulf-European travel hopes World Health Organisation has given an emergency use listing to Sinopharm Chinas Covid-19 vaccine The World Health Organisation has given an emergency use listing to Sinopharm Chinas Covid-19 vaccine making it the sixth vaccine to receive validation. Sinopharm got the WHOs approval on 7 May 2021 for emergency use giving the green light for this vaccine to be rolled out globally. The Sinopharm vaccine is approved by the following eight countries.
On 7 May 2021 the World Health Organization approved the vaccine for use in COVAX. The state-owned pharma groups move comes as concerns grow over the efficacy of conventional inactivated virus vaccines which have dominated the rollout in China. 9 hours agoTravellers to Saudi Arabia who have completed two doses of the Sinopharm or Sinovac vaccines will be accepted if they have received an additional dose of one of the four vaccines approved.
This article provides a summary of the interim recommendations. Decisions about which COVID-19 vaccines are included for example in the EU Digital COVID Certificate are taken by the EU Member StatesEMA is in charge of the scientific evaluation of vaccines for EU marketing authorisation. EMA is not involved in advising on travel requirements in the EU such as vaccination quarantine or testing for travellers.
The WHO Strategic Advisory Group of Experts SAGE on Immunization has issued Interim recommendations for the use of the inactivated COVID-19 vaccine BIBP developed by SinopharmChina National Pharmaceutical Group. Sinopharm has signed purchase agreements for 170 million doses from COVAX. Vaccines authorised in the European Union EU to prevent COVID-19 following evaluation by the European Medicines Agency EMA.
As for the Sinopharm and Sinovac vaccines they are both recognised by eight European countries. How Europe went from backwater to global dominance. Inactivated vaccines such as Sinopharms.
The other inactivated virus vaccine developed by Sinopharm is WIBP-CorV. Here is what you need to know. Walvax said last week in a stock exchange filing that it had obtained approval from the.
So Now a Chinease Vaccine called SinoPharm has approved for emergency use on 8th May 2021 its will be good opporotunity for chinas. Trenchard told Arabian Business. The Sinovac vaccine is recognised as valid proof of vaccination by these countries.
Vaccine which has been widely used across the region is expected to be passed by the WHO by the end of the week. COVID-19 Vaccines Recognised for Travel. The WHO issued its emergency use listing for the Sinopharm vaccine on May 7 2021 some 4 months after Chinas National Medical Products.
Decisions about which COVID-19 vaccines are included for example in the EU Digital COVID Certificate are taken by the EU Member States. The interim recommendations and the background. Thanks AhenselSo as of today sinopharm is accepted by greece Switzerland Austria italy Travelcorridor with uae Cyprus and Croatia if I amnot wrongJust trying to build my itenirary based on the current entry conditionsHopefully there is a travelcorridor between Germany and uae.
The imminent World Health Organisation WHO approval of Chinas Sinopharm vaccine will be a welcome relief for Middle East travellers and European economies. WHO recommends the vaccine for adults 18 years and older in a two-dose schedule with a spacing of three to four weeks. The sinopharm vaccine which has been readily used in the UAE Bahrain and other Middle East countries is awaiting official approval from the WHO.
The National Primary Health Care Development Agency on Tuesday said the National Agency for Food and Drug Administration and Control had approved Sinopharm BBIBP-CorV COVID-19 vaccine for use in. 4 hours agoVarious Vaccines like Spuntik V Russian AstraZeneca UK Covaxin India Pfizer Moderna US and other Australian American European vaccines are gets approved due to the global Pandemic.
More Eu Countries Eyeing Separate Deals With Russia For Sputnik Vaccine Politico
Eu Unveils Plans For Overseas Tourists To Return Bbc News
Eu S Covid Travel Pass Will Let Countries Choose Russia China Vaccines Politico

Czech Republic To Get China S Sinopharm Virus Vaccine
Hungary First In Eu To Innoculate With China S Sinopharm Vaccine Cgtn
Eu Slams Russia And China For Western Vaccines Disinformation Campaign Euronews
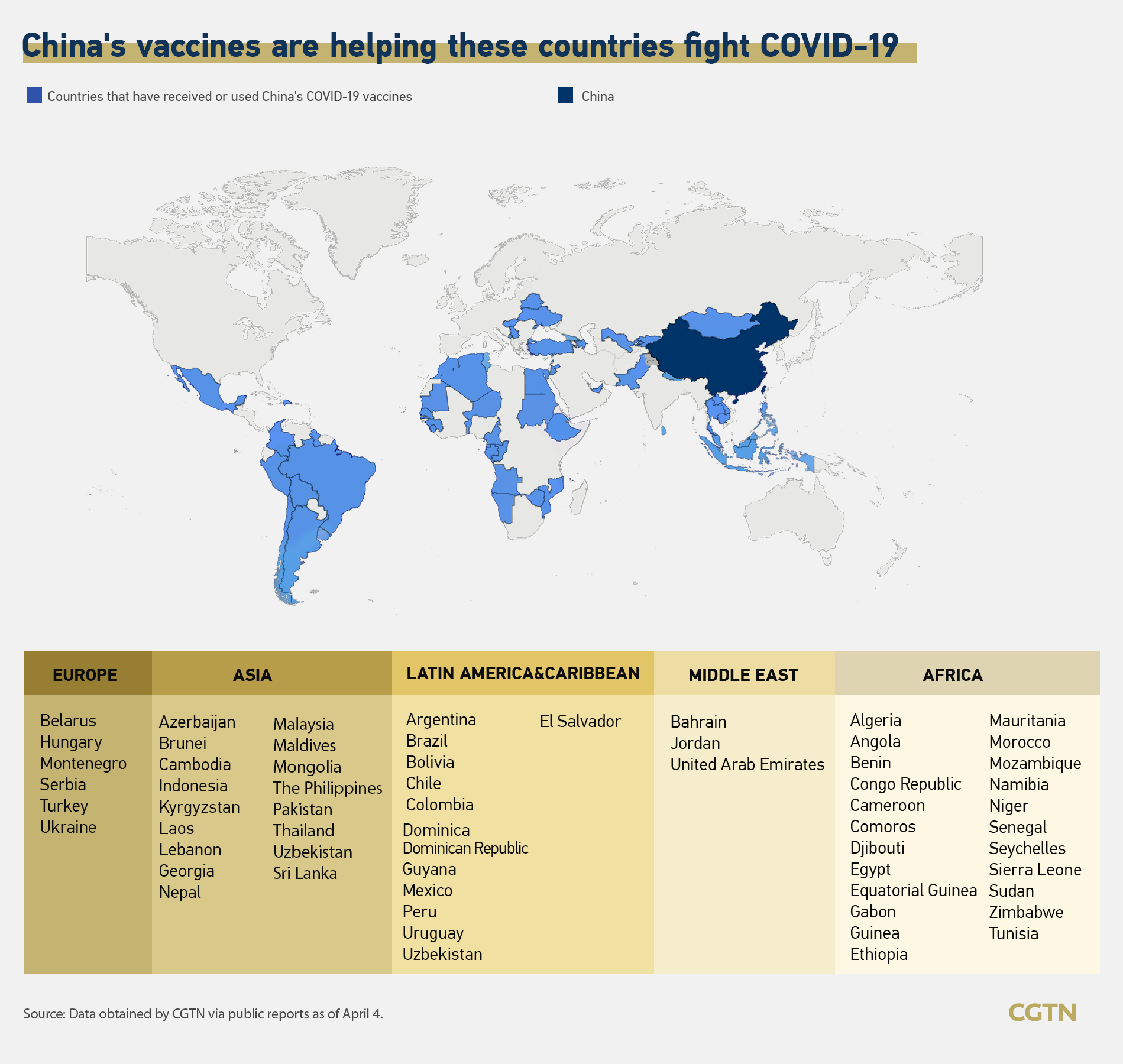
Covid 19 China Vaccines Aid More Nations With 1st Eu Gmp Certificate Cgtn

Who Approves Chinese Covid Vaccine For Emergency Use Worldwide Voice Of America English

Covid 19 Morocco To Import Millions Of Doses Of Sinopharm Vaccine Africanews

China S Vaccine Map Countries Using Chinese Vaccines Cgtn

Who Approves A Covid 19 Vaccine From China S Sinopharm For Emergency Use
China Helps Serbia Surge Ahead In European Vaccine Race
The Logic Of China S Vaccine Diplomacy Chinaobservers

Hungarians Receive Chinese Covid Vaccine As Orban Breaks With Eu Strategy Financial Times
Cansino Granted Gmp Certificate From Hungary After Sinopharm Increasing Eu Confidence In Recombinant Covid 19 Vaccine Global Times
In Eu First Sinopharm S Coronavirus Vaccine Approved By Hungary Nikkei Asia

Who And Ema Approved Vaccine List Which Covid 19 Vaccines Are On Who And Ema List Updated 30 August 2021 Wego Travel Blog

Eu Travel Covishield Sinopharm Sinovac Vaccines Are Most Widely Accepted By Eu Countries After Those Authorised By Ema Schengenvisainfo Com

Serbia To Start Producing Chinese Sinopharm Vaccine Euractiv Com
